Our end-to-end workflow turns raw target data into Phase-3–ready assets. Proprietary AI finds the hits, our labs optimise and de-risk them, and a dedicated clinical unit drives the best candidates through trials—compressing timelines and cost.
300 M-compound virtual screen delivers a ranked shortlist of hits in five days.
Medicinal chemistry and full toxicology panels refine potency and minimise risk before IND.
In-vivo efficacy and dosing studies build the data package for regulatory submission
A dedicated team advances top candidates through Phase 1-2, accelerating approval.

With access to advanced instrumentation and data analytics, our laboratory stays at the forefront of scientific progress. Our research solutions are as innovative as they are accurate - designed to keep you ahead
Amino acid sequence, 3D structure, physico-chemical properties of binding sites
Nucleotide sequence, 3D structure, genetic context (regulatory, coding, non-coding etc.)
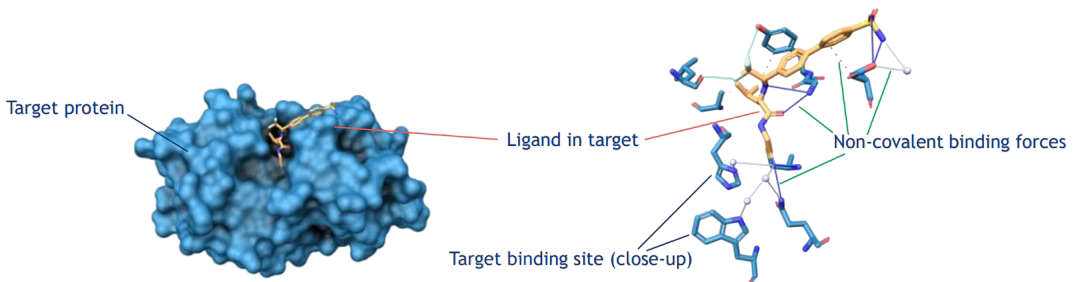
With access to advanced instrumentation and data analytics, our laboratory stays at the forefront of scientific progress. Our research solutions are as innovative as they are accurate - designed to keep you ahead
Standardised mathematical description
Unique, structure-invariant 3D fingerprint
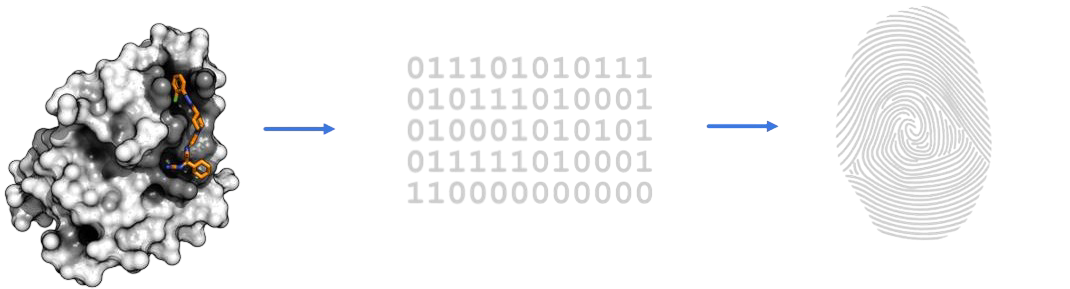
Chemical space with 300 million+ theoretical and existing compounds (small molecules)

In collaboration with ABX-CRO advanced pharmaceutical services Forschungsgesellschaft, Germany.
In-vivo efficacy, PK/PD, and full GLP toxicology studies generate an IND-ready data package.
First-in-human safety & PK (Phase 1) followed by proof-of-concept efficacy and dose-optimisation (Phase 2) in multi-centre trials run with ABX-CRO.

Outperforming competitive in silico methodologies quantitatively (hit rate) and qualitatively (affinity) by a Factor of 10x
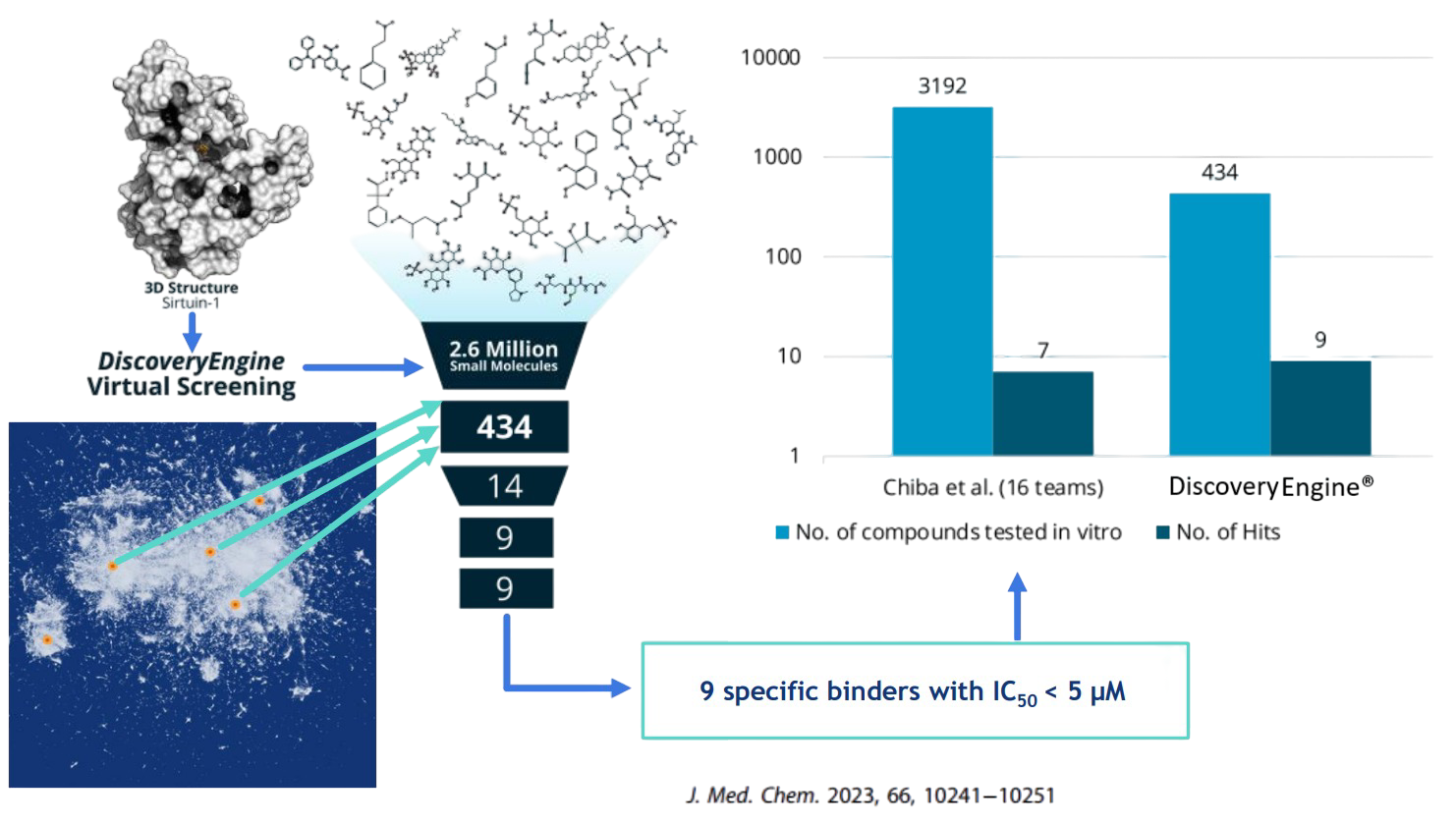
PharmAI Discovery and its daughter company PharmAI has successful collaboration projects with more than 20 prominent actors within the field.
